The therapy, called Otarmeni, made by Regeneron Pharmaceuticals was approved by United States Food and Drug Administration under National Priority Voucher Program.
Key Highlights of the Approved Therapy
- OTOF Gene Mutation: Therapy is designed to treat hearing loss caused by OTOF gene mutations.
- OTOF Gene codes for a protein called otoferlin, needed by inner ear cells to translate vibrations into signals interpreted by brain.
- When people carry two defective copies of this gene (one from each parent), it causes severe-to-profound hearing loss, accounting for 2%- 8% of inherited, non-syndromic cases.
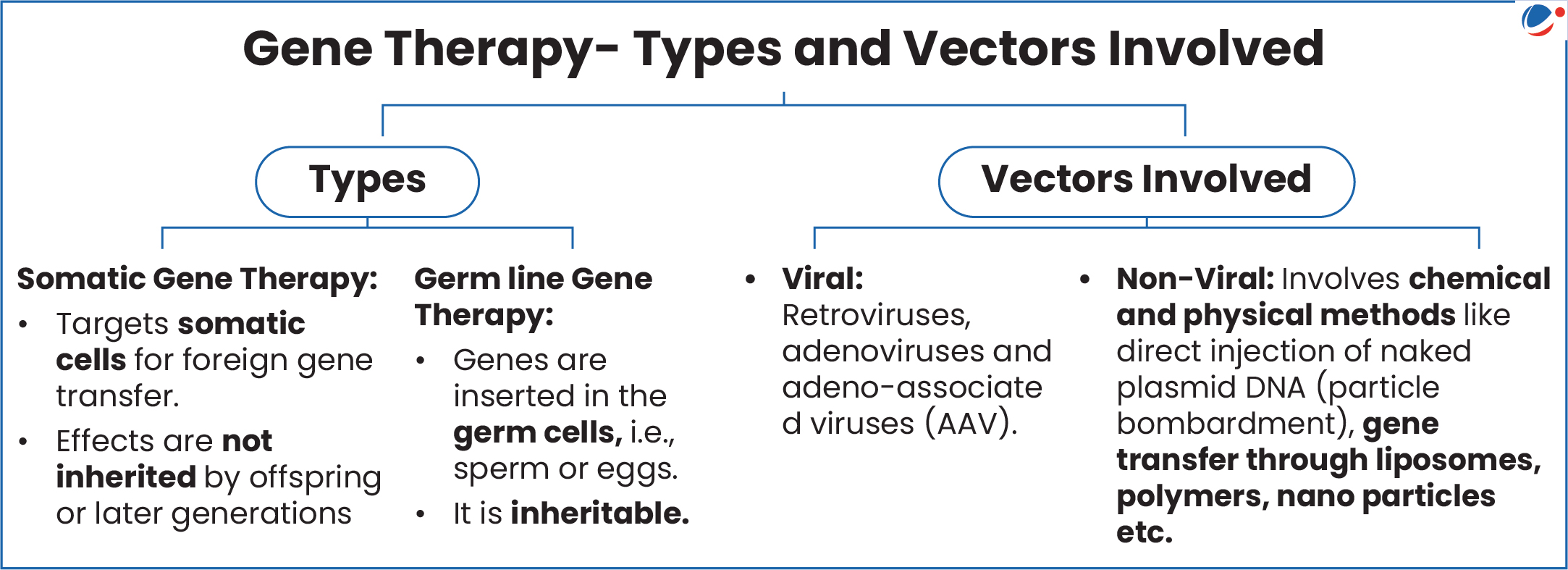
- Otarmeni: Is a one-time biologic-device combination product including a dual adeno-associated virus serotype 1 (AAV1) vector gene therapy.
- It delivers a functional copy of OTOF gene to inner hair cells to restore otoferlin production and auditory signalling.
- Dual adeno-associated virus (AAV) therapy, involves the use of two different adeno-associated viruses (AAVs) to deliver therapeutic genes.
About Gene Therapy
- Meaning: Refers to a collection of methods that allows correction of a gene defect. It involves insertion of genes into a person’s cells and tissues to treat a disease.
- A vector is a carrier used to deliver the therapeutic gene into target cells.